CranioSim
Within this project, the aim is to develop a surgical simulator for cranial grafts harvesting techniques.
Motivation
The first transplantation of a bone graft was reported in 1668, by the Dutch surgeon Job van Meekeren, who transplanted a graft harvested from a dog’s skull into a soldier’s cranial bone [1]. Nowadays, bone grafts are harvested from distant sites, like the iliac crest, the tibia, maxilla, mandible, ribs, sternum and from the calvarial bone [2]. Graft harvested from the calvarial bone has become a quite popular site for donoring autologuous grafts, especially for grafts used in cranio-maxillo-facial surgery [3].
In 1982, the first use of autogenous calvarial bone as a graft material in face reconstruction was reported by Tessier [4]. The cranial bone offers numerous advantages as graft material, which include a reduction of post-operative pain of the donor site, a well hidden scar beneath the scalp, a decreased hospitalisation time, and in case of maxilla-facial surgery, a single surgical field. Bone grafts from the skull undergo less resorption and may revascularise more rapidly [5].
Due to the reason, that the skull is not flat but convex, the gathering of a passable transplant of the outer table is not that easy. During the harvest, it is absolutely necessary to avoid an entering of the skull’s vault [3]. The disadvantage of this donor site may lie within the risk of a dural tear, contour deformity of the skull and only a minimal amount of cancellous bone which is available for harvest [2].
   |
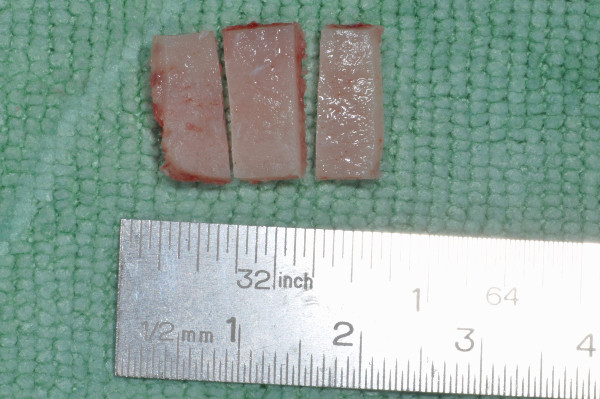   |
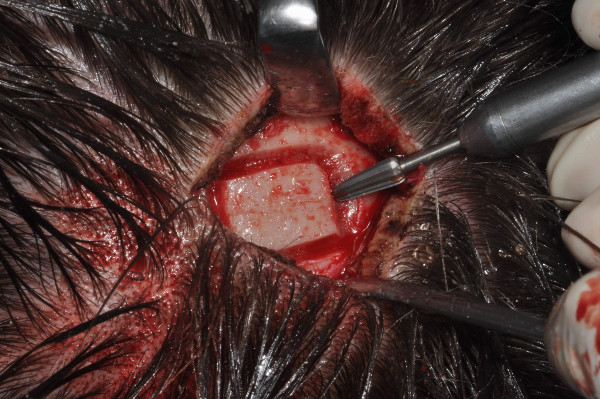
| Figure 1: Preparation for graft extraction (Prim. Malek, AKH Linz). | Figure 2: Extracted graft (Prim. Malek, AKH Linz). |
The skull vault’s bone consists of three layers; the outer and inner table (tabulae externa and interna) consist of compact bone whereas the middle layer, called diploe, consists of spongy, trabecular bone . The preferred harvest site for calvarial grafts is the parietal bone, because of its thickness [5].
A typical technique (see Figure 3) to harvest calvarial bone is performed as follows:
after the scalp is cut open and held open with clamps, the area around the area which will be harvested is cut with a cutting burr. The depth of this cut depends on the thickness of the calvarial bone. Then, an oscillating saw can progress below the diploic space. As last step, the final cut is performed with a curved osteotome, which cuts the last diploic connections between the outer and inner table of the cranial bone [3] in order to obtain the autologuoues grafts (Figure 2).
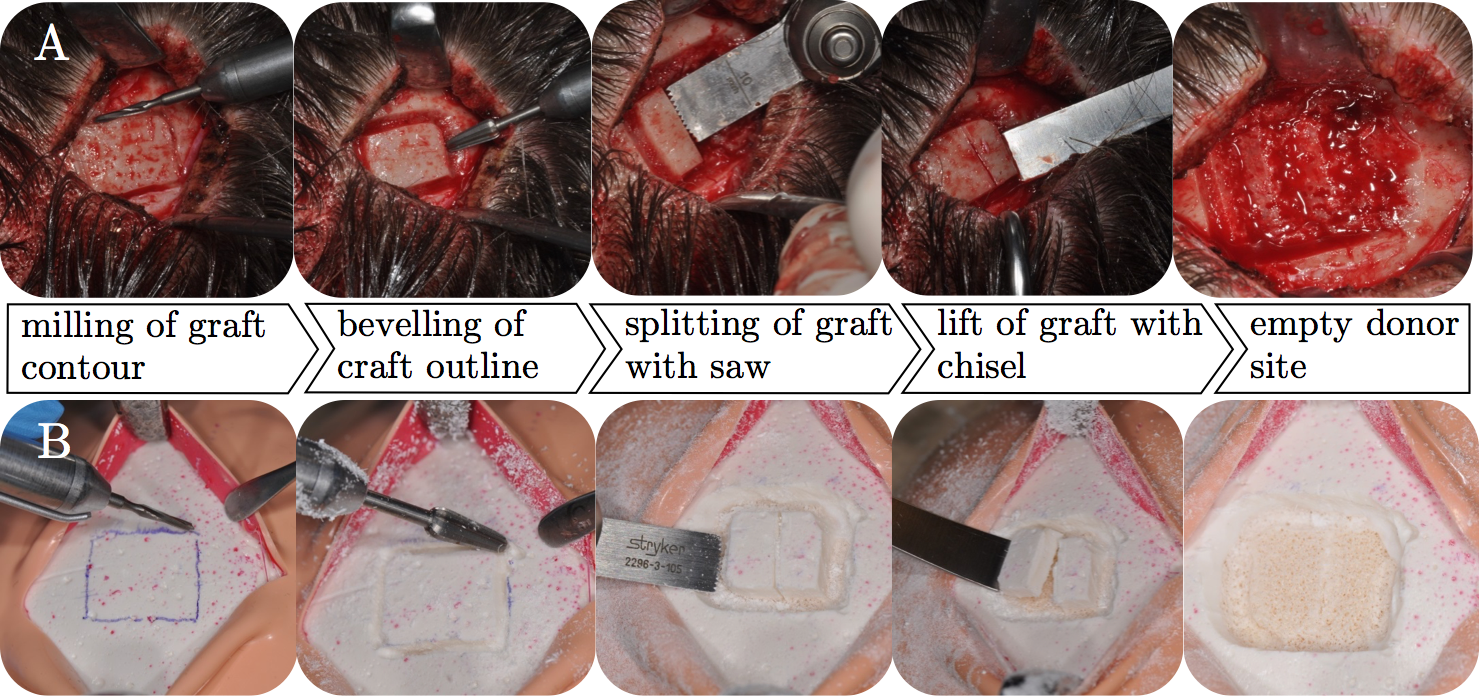   |
| Figure 3: Procedure for harvesting calvarial bone. A – patient, B – simulator. |
Simulator
Artificial Cranial Bone
In order to set up a simulator for parietal graft lift training, polyurethane (PU)-based bone surrogate composites (Figure 4) enriched with mineral fillers, were developed [6, 7, 8]. In order to mimic a spongy bone covered by a cortical layer, thus resembling the composition of human skull, four different material compositions were created. To create an open cell cancellous structure, the PU resin was supplemented with water as a blowing agent and a cell stabiliser. This cancellous PU mixture was further supplemented with two fillers- calcium phosphate (CaP, Ca3(PO4)2) and calciumcarbonate (CaC, CaCO3)- in amounts of 50 (CaP050, CaC050) and 100% (CaP100, CaC100) of the whole mass to vary structure and hardness. After curing, the cancellous bone surrogate blocks were cut with a band saw achieving a flat surface which was coated with a second mixture. This cortical mixture, which consisted of the PU resin and the mineral-based fillers, was smoothly spread on the cancellous bone blocks with the aid of a spatula, resulting in an even layer of 1.5 mm. Thus, the artificial cortical layer was in accordance with a human outer table, which thickness is around 1.6 ± 0.2 mm [9].
Patient Phantom
A training simulator by means of a patient phantom for harvesting cranial bone grafts was built with artificial bone surrogates. The artificial skull cap was placed on a silicone base with a brain-shaped bulge to prevent slipping. Furthermore, the skull cap was covered with a layer of artificial soft tissues (muscles and skin). After a skin incision and the retraction of the soft tissues, the surgeon is able to train the same procedural steps as in a real parietal graft lift surgery. The simulator prototypes were tested by two experienced surgeons (see Figure 5).
 |
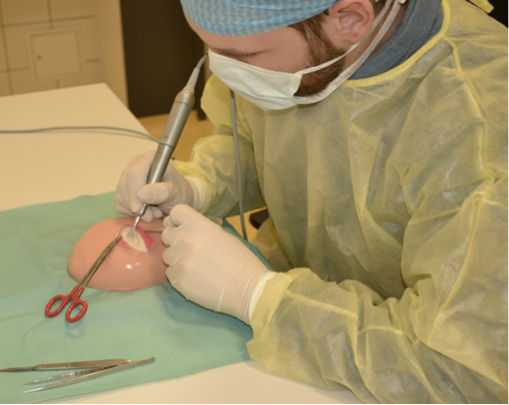   |
| Figure 4: Artificial skull cap of the simulator showing cross-sections of artificial bone. | Figure 5: Surgeon training on the simulator with real instruments. |
The following movie shows a surgeon extracting a cranial graft.
Validation
The simulator, represented by the customised patient phantom was validated into two directions: by means of characteristic parameters for drilling, milling and sawing by comparison with human parietal bone and due to an evaluation study gaining face, content and constructive validity measures.
Surgical machining
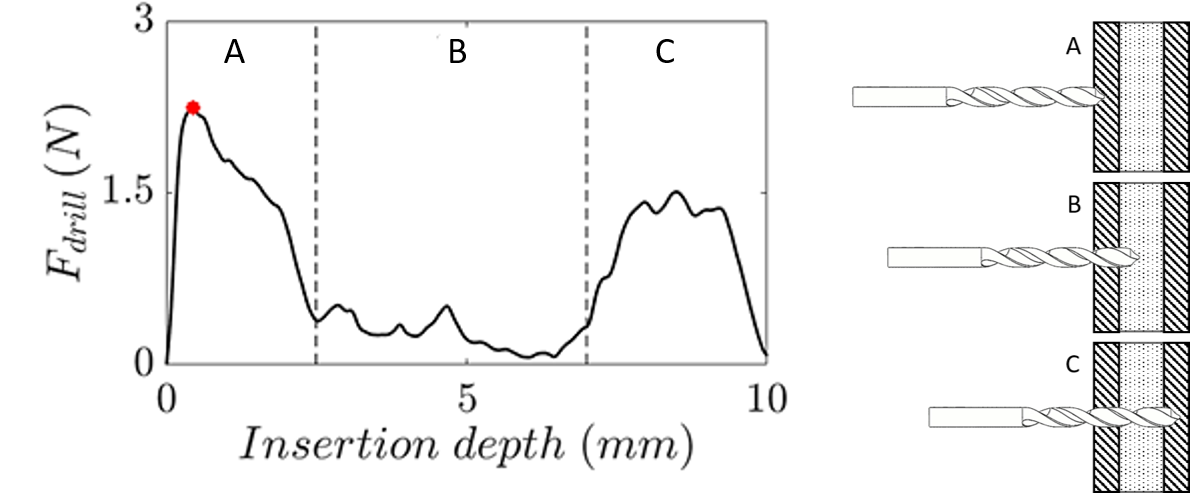
In accordance with the surgical procedure of a split thickness tabula externa graft lift the maximum puncture forces of the drill- and mill heads penetrating the tabula externa were used to compare the machining characteristics of artificial and human skull bones (see Figure 6). Furthermore, the maximum force of the cancellous sawing procedure, which is due to a full sawblade insertion into the cancellous bone, was identified as a characteristic parameter.
   |
||
| Figure 6: Drilling force over insertion depth at different positions: A: tabula externa, B: diploe, C: tabula interna. |
Drilling
The maximum drilling forces in axial direction during the investigation of the testing blocks were recorded for CaC050 bone surrogates (2.5 ± 0.2 N). In contrast, the recorded forces during the drilling of the human specimens were 1.8 ± 0.5N. The artificial bone materials had 18% (CaP050), 20% (CaP100), 42% (CaC050) and 32% (CaC100) higher average maximum drilling force than the tested human bones. The Shapiro-Wilk test showed normal distribution for all drilling measurement results and homogenous variances were determined for all groups. Significant differences between the human specimens and the bone surrogates were not detected (see Figure 7).
Milling
The lowest axial milling forces were detected for surrogate CaC050 (1.3 ± 0.1 N) which were clearly lower than the milling forces observed for human parietal bones (1.7 ± 0.3 N). However, the mean maximum milling forces were 18% (CaP050), 21% (CaP100), 25% (CaC050) and 16% (CaC100) smaller than the results of human specimens. The Shapiro-Wilk test detected a non-normal distribution of the human specimen. Because of this a Mann-Whitney-U-test was used to detect differences between human and artificial specimens. All bone surrogates revealed high p-values (see Figure 7).
Sawing
The Shapiro-Wilk test indicated normal distribution for all sawing measurements and the Levene-test showed homogenous variances. Thus, an unpaired Student‘s -t test was used to detect differences between human and artificial bones. The the lowest sawing forces were found for CaC050 (0.5 ± 0.1 N). Thus, the mean maximum sawing forces of the artificial bones CaP050, CaP100, CaC050, CaC100 were 32%, 11%, 43%, 26% smaller than the human bone samples (see Figure 7).
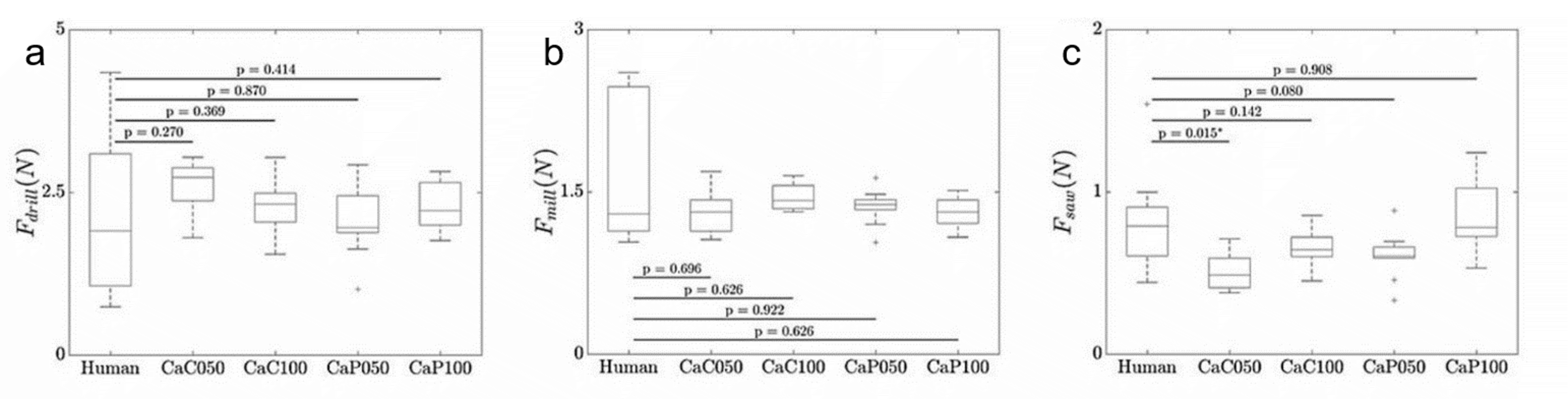   |
| Figure 7: Milling-, drilling- and sawing forces for artificial materials in comparison to human parietal bone. |
Anatomical layers
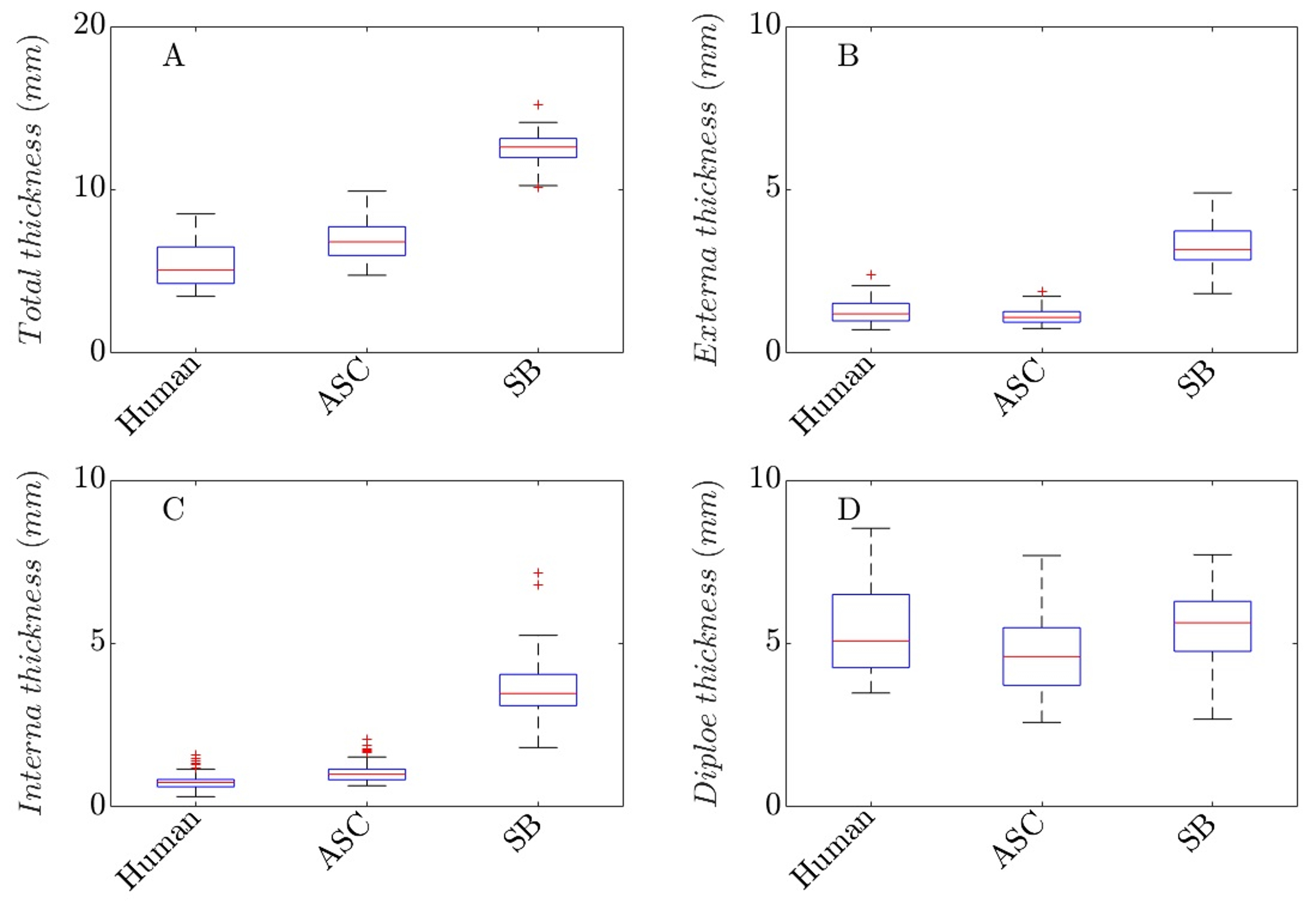
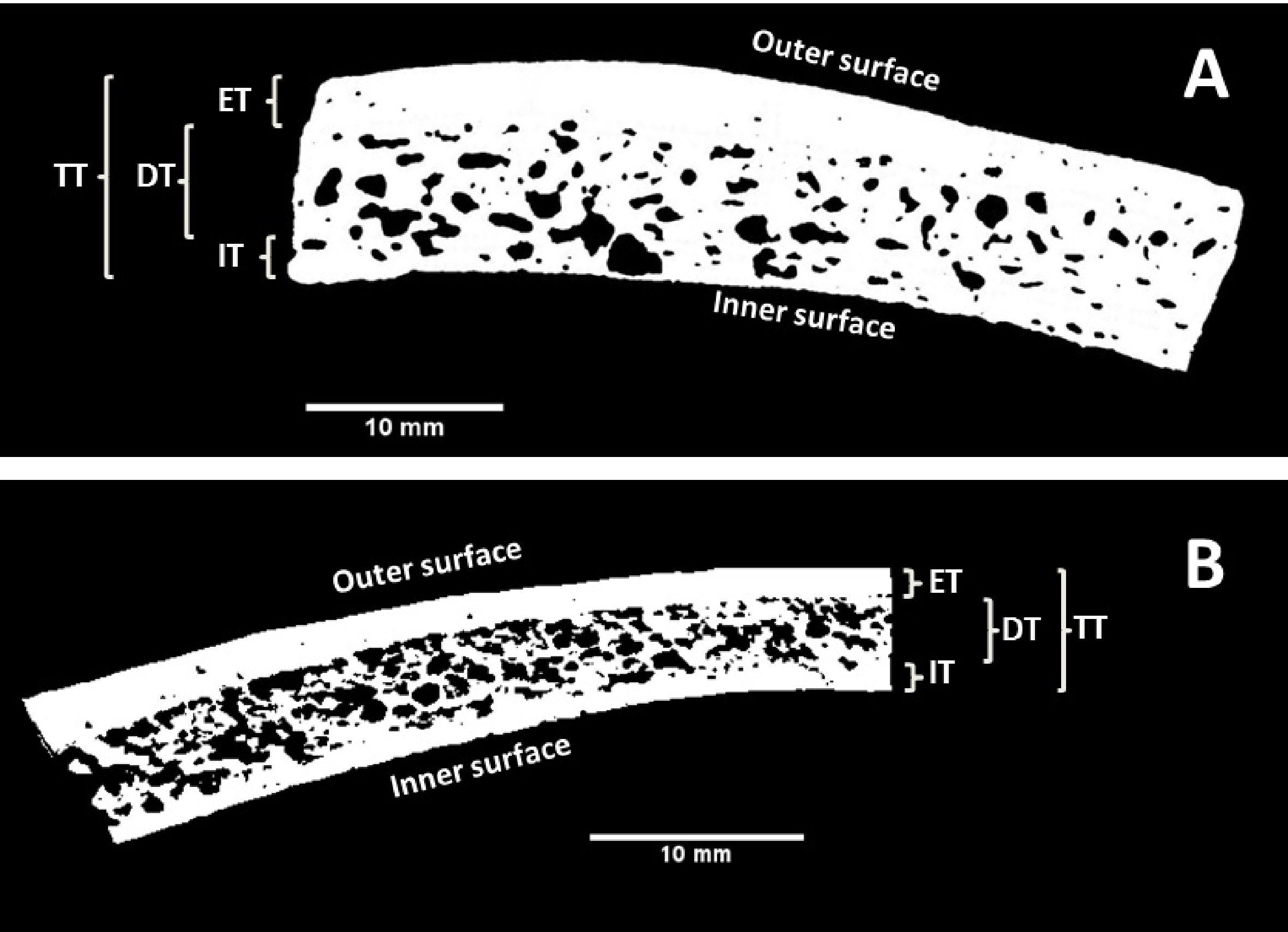
To create an artificial skull cap with anatomically realistic bone layers, the bone layer thickness values of the tabula externa, diploe and tabula interna as well as the total thickness of the parietal bone samples were investigated (see Figure 8). The results were used as a reference for the development of the bone layers of the artificial skull cap (ASC), especially for the estimation of the amounts of materials needed to mold the bone layers. All human specimens were scanned with a micro-computed tomography (CT80, Scanco 41 Medical, Brüttisellen, Switzerland). For all images, resolution preferences of 70 kV, 114A and a 200ms integration time were used resulting in a slice thickness of 0.09mm.
For all samples, the thickness of the inner and outer cortical layers as well as the total thickness were measured. Hence, the thickness of the diploic space could be calculated by subtracting the cortical layer thickness values from the total bone thickness.
   |
||
| Figure 8: A – cross section of a human parietal bone sample, B – artificial bone. ET/IT – tabula externa/interna thickness, DT – diploe thickness, TT – total thickness. |
The results are summarised by Figure 9. Human bone samples showed a total bone thickness of 5.31 ± 1.24mm. The total thickness of the artificial skull cap (ASC) was 6.94 ± 1.20mm and thus were 30.1% larger than the human total thickness. The Sawbones (SB) skull samples had a total thickness of 12.51 ± 1.14mm and though were more than twice as thick (+135.4 %) as the human bone samples. The externa thickness values of the human bones were 1.26 ± 0.34mm. The artificial skull caps were similar (1.15 ± 0.25mm) and only were 9 % larger than the human tabula externa while the SB samples were more than 165 % larger (3.35 ± 0.73mm).
Similar results were found for the interna thickness values. The average interna thickness of the human parietal bone was 0.76 ± 0.22mm. ASC (1.07 ± 0.34mm, +40.7%) and SB (3.69 ± 1.18mm, +385.5%) were clearly thicker.
Validation study
The aim of this study was to verify the face, content and construct validity of the novel model-based simulator within a validation study [10]. Participants were enforced to perform a tabula externa graft lift during a training session on the simulator. Task performance was analysed with a standardised assessment tool.
Prior to the training task, the participants (n=15) completed questionnaires based on demographics and their medical experiences. Then, experts and novices were invited to perform lifts of a 2×1.5cm graft split in three equal proportions from the parietal tabula externa using the model-based simulator.
The study-participants rated the realism and appropriateness as a teaching modality of the TabSIM along a 5-point Likert scale. The realism, or face validity, was confirmed with an average score of 4.2 of a maximum of 5 points. Likewise, the content validity, which proved the appropriateness of the simulator as a teaching modality, was proven with an average score of 4.8 of maximum 5 points. During the evaluation of task performance, the expert surgeons were evaluated significantly better than the novices, which proved the construct validity of the simulator (see Figure 10).
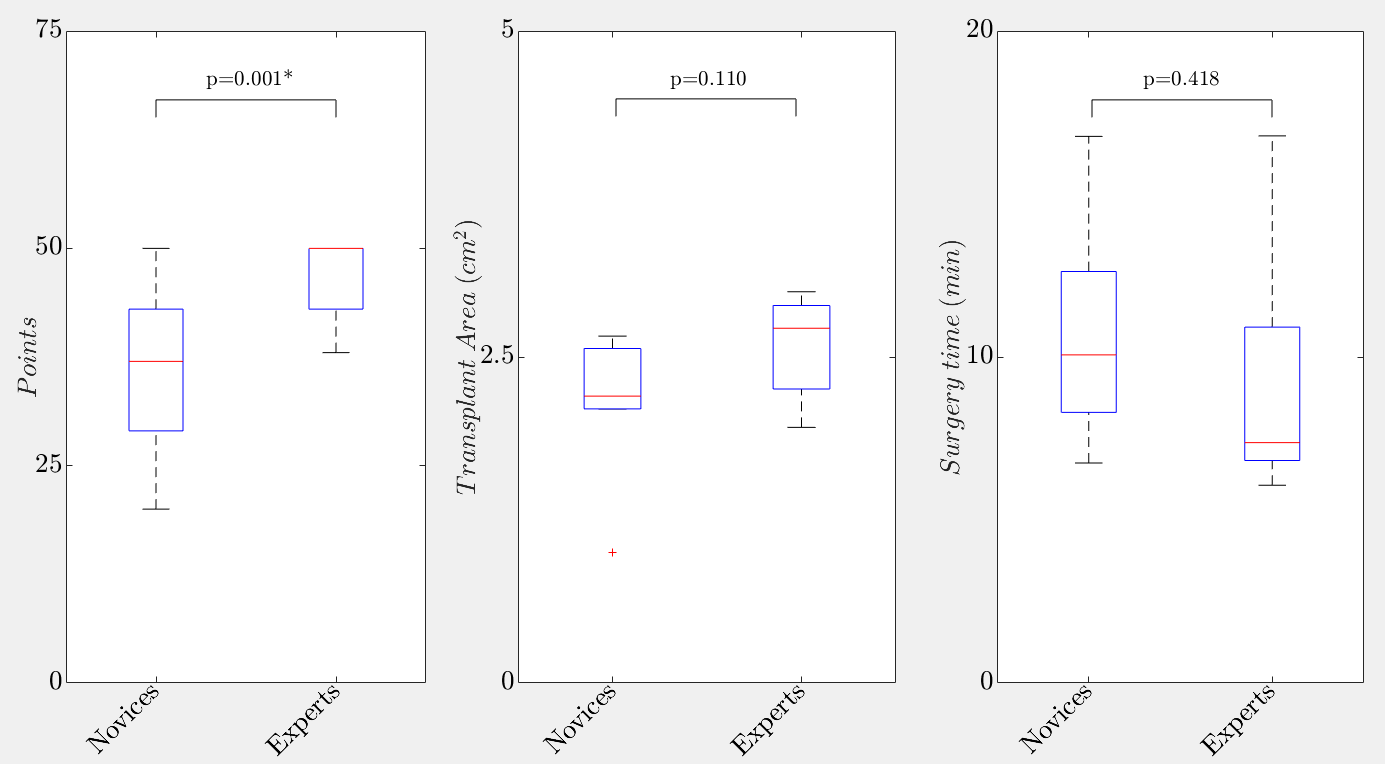   |
| Figure 10: Boxplots showing the differences between novices and experts for the global rating scale, the transplant area and the surgery time. |
[Bibtex]
@Article{DeBoer1988,
title = {The history of Bone Grafts},
author = {Hermann H. De Boer},
journal = {Clinical Orthopaedics and Related Research},
year = 1988,
pages = {292-298},
volume = 226,
owner = {p41066},
timestamp = {2014.07.01}
}[Bibtex]
@Article{Zouhary2010,
author = {Zouhary, Kenneth J.},
title = {Bone Graft Harvesting From Distant Sites: Concepts and Techniques},
journal = {Oral and Maxillofacial Surgery Clinics of North America},
year = {2010},
volume = {22},
number = {3},
pages = {301-316},
month = {Aug},
issn = {1042-3699},
doi = {10.1016/j.coms.2010.04.007},
owner = {p41066},
publisher = {Elsevier BV},
timestamp = {2014.07.01},
url = {http://dx.doi.org/10.1016/j.coms.2010.04.007},
}[Bibtex]
@Article{Yoshimura1990,
title = {An instrument for harvesting the outer table of the skull},
author = {Yoshimura, Yohko and Nakajima, Tatsuo and Onishi, Kiyoshi},
journal = {Journal of Cranio-Maxillofacial Surgery},
year = 1990,
month = {May},
number = 4,
pages = {179-181},
volume = 18,
doi = {10.1016/s1010-5182(05)80516-5},
issn = {1010-5182},
owner = {p41066},
publisher = {Elsevier BV},
timestamp = {2014.07.01},
url = {http://dx.doi.org/10.1016/S1010-5182(05)80516-5}
}[Bibtex]
@Article{Tessier1982,
title = {Autogenous bone grafts taken from the calvarium for facial and cranial applications},
author = {P Tessier},
journal = {Clin Plast Surg},
year = 1982,
pages = {531-538},
volume = 9,
owner = {p41066},
timestamp = {2014.07.01}
}[Bibtex]
@Article{Bruno1994,
author = {Bruno, Barbara J. and Gustafson, Patricia A.},
title = {Cranial Bone Harvest, Grafting},
journal = {AORN Journal},
year = {1994},
volume = {59},
number = {1},
pages = {242-251},
month = {Jan},
issn = {0001-2092},
doi = {10.1016/s0001-2092(07)65324-1},
owner = {p41066},
publisher = {Elsevier BV},
timestamp = {2014.07.01},
url = {http://dx.doi.org/10.1016/S0001-2092(07)65324-1},
}[Bibtex]
@Article{Hollensteiner2017d,
Title = {{Novel bone surrogates for cranial surgery training}},
Author = {Marianne Hollensteiner and David Fürst and Benjamin Esterer and Peter Augat and Falk Schrödl and Stefan Hunger and Michael Malek and Daniel Stephan and Andreas Schrempf},
Journal = {{Journal of the Mechanical Behavior of Biomedical Materials}},
Year = {2017},
Abstract = {Abstract Parietal graft lifts are trained on human or animal specimens or are directly performed on patients without extensive training. In order to prevent harm to the patient resulting from fast rotating machinery tools, the surgeon needs to apply appropriate forces. Realistic haptics are essential to identify the varying parietal bone layers and to avoid a penetration of the brain. This however, requires experience and training. Therefore, in this study, bone surrogate materials were evaluated with the aim to provide an anatomically correct artificial skull cap with realistic haptic feedback for graft lift training procedures. Polyurethane composites made of calcium carbonate and calcium phosphate were developed and were used to create customized bone surrogates, imitating both cancellous and cortical bone. Mechanical properties of these surrogates were validated for drilling, milling and sawing by comparison with human parietal bones. For that, surgical tool tips were automatically inserted into artificial and human bones in a customized test bench and the maximum axial insertion forces were analyzed. Axial tool insertion measurements in human parietal bones resulted in mean maximum forces of 1.8±0.5N for drilling, 1.7±0.3N for milling and 0.9±0.1N for sawing. Calcium carbonate-based materials achieved higher forces than the human bone for drilling and milling, and lower forces for sawing. The calcium phosphate-based bone surrogates showed comparable axial insertions forces for all investigated tools and were identified as a suitable surrogate for drilling (p=0.87 and 0.41), milling (p=0.92 and 0.63) and sawing (p=0.11 and 0.76) of the cortical layer and the cancellous bone, respectively. In conclusion, our findings suggest, that a suitable material composition for artificial parietal bones has been identified, mimicking the properties of human bone during surgical machinery procedures. Thus, these materials are suitable for surgical training and education in simulator training.},
Volume = {72},
Number = {Supplement-C},
Pages = {9 -- 51},
Url = {http://www.sciencedirect.com/science/article/pii/S1751616117301741}
}[Bibtex]
@Article{Hollensteiner2017c,
Title = {Development of parietal bone surrogates for parietal graft lift training},
Author = {Marianne Hollensteiner and David Fuerst and Benjamin Esterer and Stefan Hunger and Michael Malek and Peter Augat and Falk Schroedl and Daniel Stephan and Andreas Schrempf},
Journal = {{Current Directions in Biomedical Engineering}},
Year = {2016},
Url = {https://doi.org/10.1515/cdbme-2016-0140},
Abstract = {Currently the surgical training of parietal bone graft techniques is performed on patients or specimens. Commercially available bone models do not deliver realistic haptic feedback. Thus customized parietal skull surrogates were developed for surgical training purposes. Two human parietal bones were used as reference. Based on the measurement of insertion forces of drilling, milling and saw procedures suitable material compositions for molding cortical and cancellous calvarial layers were found. Artificial skull caps were manufactured and tested. Additionally microtomograpy images of human and artificial parietal bones were performed to analyze outer table and diploe thicknesses. Significant differences between human and artificial skulls were not detected with the mechanical procedures tested. Highly significant differences were found for the diploe thickness values. In conclusion, an artificial bone has been created, mimicking the properties of human parietal bone}
}[Bibtex]
@Article{Hollensteiner2017b,
Title = {Novel Simulator for Cranial Graft Lift Training},
Author = {Marianne Hollensteiner and David Fuerst and Benjamin Esterer and Stefan Gabauer and Peter Augat and Falk Schroedl and Andreas Schrempf},
Journal = {{Proceedings of the IAUP Triennial Conference}},
Year = {2017},
Abstract = {Cranial grafts are used for the reconstruction of skeletal defects after trauma, tumor, infection or congenital pseudarthrosis [1] and due to their biocompatibility are more favored [2]. A common method to harvest parietal grafts is the split thickness graft method which is characterized by fast rotating surgical drives. To avoid accidents, appropriate thrust forces and drilling speeds have to be applied. Thus extensive training of novice surgeons is necessary. One training modality are model simulators, which provide a physical phantom with realistic anatomy and haptic feedback. Studies have shown that, in contrast to visual guided training only, simulators with haptic feedback have higher surgical skill-transfer to novel surgeons [3]. The aim of this study was to validate artifi cial parietal bones for graft lift training. Structural parameters of all bony layers were measured and characteristic forces during surgical machinery procedures were recorded [4]. Further, a simulator prototype was developed and tested by two experienced surgeons.}
}[Bibtex]
@Article{Jung2003,
author = {Jung, Y-S and Kim, H-J and Choi, S-W and Kang, J-W and Cha, I-H},
title = {Regional thickness of parietal bone in Korean adults.},
journal = {International journal of oral and maxillofacial surgery},
year = {2003},
volume = {32},
pages = {638--641},
month = dec,
issn = {0901-5027},
__markedentry = {[A20887:6]},
abstract = {To clarify the clinical utility of the parietal bone graft in maxillofacial reconstruction, we performed an anatomical study by measuring the regional thickness of the parietal bone in 47 Korean adult dry skulls. Before sectioning of the calvaria, the appropriate anatomical landmarks were marked on each specimen. We measured the total thickness of the parietal bone, and the thickness of the outer and inner cortical plates at various points in each section of parietal bones using a digital caliper under the stereomicroscope. The total thickness of the parietal bone ranged from 5.04mm to 7.17mm, and there was no statistical difference in the total thickness of the parietal bone on the same points bilaterally. The parietal bone tended to be thicker toward the lambda point than at the coronal suture area. On the other hand, the outer plate of parietal bone was thickest at the point nearest to the coronal suture, and the inner plate proved thickest at the posteromedial area. In conclusion, this study showed that the better donor site of the parietal bone for maxillofacial reconstruction is located at its more posterior and medial area.},
citation-subset = {D, IM},
completed = {2004-02-04},
country = {Denmark},
created = {2003-11-25},
doi = {10.1054/ijom.2002.0415},
issn-linking = {0901-5027},
issue = {6},
keywords = {Adult; Analysis of Variance; Bone Density; Bone Transplantation; Humans; Parietal Bone, anatomy & histology, transplantation; Reference Values; Tissue and Organ Harvesting},
nlm-id = {8605826},
owner = {NLM},
pii = {S0901-5027(02)90415-7},
pmid = {14636616},
pubmodel = {Print},
pubstatus = {ppublish},
revised = {2006-11-15},
timestamp = {2017.10.04},
}[Bibtex]
@Article{Hollensteiner2017e,
Title = {{Validierung eines modell-basierten Simulators für das Training in der CMF Chirurgie}},
Author = {Marianne Hollensteiner and Michael Malek and Stefan Hunger and David Fuerst and Benjamin Esterer and Peter Augat and Falk Schroedl and Daniel Stephan and Andreas Schrempf},
Journal = {{Proceedings of the 3rd Kepler Science Day}},
Year = {2017},
Abstract = {Das Heben von Tabula Externa Transplantaten aus der parietalen Schädelkalotte ist eine anerkannte Praxis zur Behandlung von knöchernen Deformitäten. Um Risiken zu minimieren, müssen während des chirurgischen Eingriffs entsprechende Kräfte und Geschwindigkeiten der chirurgischen Instrumente angewendet werden. Um diesen Eingriff zu trainieren, wurde ein modell-basierter Simulator entwickelt. Um zu beweisen, dass dieser neue Simulator für das Training und die Ausbildung von Novizen geeignet ist, wurde eine Validierungsstudie im Kepler Universitätsklinikum Linz durchgeführt.},
Volume = {},
Number = {},
Pages = {},
Url = {}
}